|
|
Post by Katharina Sugiarti on Feb 8, 2008 10:18:04 GMT 7
Kira2 .... Murahan mana biaya listrik dengan cara beli batu es balok setiap harinya?
|
|
|
|
Post by rajah on Feb 8, 2008 10:55:09 GMT 7
Pak Bcal kreatif euy....jadi idenya memperpanjang pipa tembaga itu sampai ke luar kulkas ya?
Itu kulkas kecil yang pake freon kan.... mungkin kapasitasnya kompresornya lebih ideal untuk mendinginkan udara di ruang tertutup yang kecil, tapi ....menarik juga membandingkan waktu pendinginan air dibanding kecepatan berkurangnya air dalam wadah. Pipa tembaga itu belinya di mana dan harga berapa mr Bcal ?
|
|
|
|
Post by ∂r¹ƒ on Feb 8, 2008 11:21:12 GMT 7
pipa tembaganya di celup ke dlm air di wadah plastik kotak yg ada evaporatornya (terbenam air juga) pak rajah, tembaga kan penghantar panas yg bagus jadi air yg di wadah cat besar saya sirkulasiin pakai pompa submersib lewat pipa tembaga yg didalam kulkas trus ke wadah misting trus balik lg ke wadah cat besar, nah panasnya diserap evaporator jadinya dingin deh..
Kulkasnya pake yg dua pintu pak, saya pakai frezzernya tapi ruang di bawah frezzer udah nga ada dingin2nya sedikitpun mungkin udah dipake buat dinginin air di wadah plastik kotak yg di frezzer..
Pipa tembaganya saya beli di toko peralatan AC, kalo harganya tergantung diameter pipanya, yg saya pakai diameter 5/18" harganya 15 rb per meter..
|
|
|
|
Post by rajah on Feb 8, 2008 11:50:23 GMT 7
Gambar yang paling atas dan yang ketiga dari atas itu bagian dalam freezer ya? Jadi pipa tembaganya tetap di dalam freezer tapi dilingkar-lingkarin dan direndam di air yang ada di wadah cat itu ?
Terus evaporatornya kenapa direndam juga boss, emang evaporator tuh bentuknya kaya gimana, dan posisi sebelumnya di mana (kalo ga salah di belakang kulkas kan?). Maap kalo banyak nanya ;D
|
|
|
|
Post by ijul on Feb 8, 2008 12:07:41 GMT 7
Murah mana sama kulkas biasa, yg bagian pintunya dicopot, lalu disambungkan langsung ke greenhouse? Kulkas terbuka tadi, langsung ditempelkan ke pintu greenhouse. Sisa bodi kulkas lainnya diluar. Greenhousenya jangan gede2 ah... Ini prinsipnya hanya "memperbesar kulkas"  |
|
|
|
Post by syx on Jul 7, 2008 9:43:27 GMT 7
There are some fairly common substances that make good refrigerants. Propane is one of them and is classified as R 290. The ban on R 12 greatly intensifies the fascination of being able to use ordinary propane, from the hardware store, as a refrigerant in a home built experiment. Sources say that propane is compatible with the mineral oil that is used in R-12 systems. 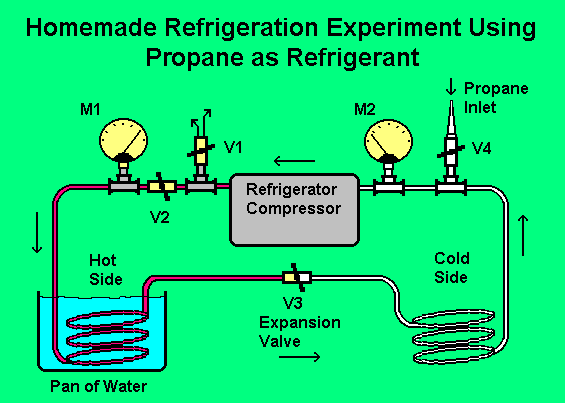  The picture shows how it was built. The 15 foot length of copper tubing was chosen arbitrarily and cut in the middle; a valve (V3) was inserted to be used as the expansion valve. This is the valve that takes the place of the normally used capillary tube. The valve has to be adjusted all the time as the temperature changes and is quite critical. The adjustment is easy though because of the two meters installed. The reason for having the other three valves is to make the system capable of evacuating itself prior to the addition of the propane. The picture is mostly self explanatory and a list of operation procedures is given below. Operating Procedure1. Start by evacuating the system. V1 and V3 open, V2 and V4 closed. Run the pump for a few minutes. At first air will be exiting from V1. After a while it will diminish to nothing. The meter that I used for M2 happened to be the type that also reads vacuum. The system evacuated to below 20". 2. Close V1 and open V2. The system is now running with most of the air removed. Connect the propane cannister through a high pressure rubber hose to the inlet barb on V4. Having a fitting on the cannister with a valve, as described above, is helpful. Open V4 and the valve on the propane cannister until both meters read about 70 or 80 psig. V4 and the valve on the propane cannister can now be closed and the rubber hose removed from the barb on V4. The system is now charged. 3. Close V3 most of the way until you get a reading on M2 of between 15 and 20 psig. This seems to be the optimal settings on my system. M1 should rise to 200 or more psig. Within seconds, the copper tube on the low pressure side of V3 should start to frost up. As the system runs, you will need to keep on top of the pressure setting on the meters by adjusting V3. The system has been a thrill to operate in spite of this. Maybe later, I will find an improved form of V3; it is a very crude expansion control. After a while, the water that is cooling the hot side will get warm and the efficiency of the system will diminish, making a change of water desirable. The psig pressure readings given for the meter readings above, mean pressure in pounds per square inch as compared to atmospheric pressure. This is called gauge (g) scale. Pressure readings sometimes are given as psia. This means pressure in pounds per square inch as compared to an absolute (a) vacuum. Thus, the difference between a reading listed as psig and one listed as psia would be roughly 14 to 15 psi. The psia reading would be higher because it includes atmospheric pressure. What is happening?Propane is compressed by the compressor into the hot side or high pressure side tubing. The propane, like any gas tends to get hot. If the tubing is kept cool, the high pressure propane will condense under the pressure into a liquid. Condensing tends to make it even hotter, but the idea is to cool it down; in this case we are using water. This tubing full of room temperature liquid is now the same thing as the cannister full of propane. When the expansion valve is opened just a little, the high pressure liquid propane squirts into the cold or low pressure side tubing and vaporizes; just like it did when squirting it on to the thermometer. Any time a liquid vaporizes, it tends to absorb heat and make things, that it comes in contact with, very cold. The vapor finally finds its way to the input side of the pump where it is compressed again. Sumber : home.earthlink.net/~lenyr/refrig.htm |
|